TEST COVID-19
laboratory test of "LUIGI SACCO" HOSPITAL OF MILAN
From the experimentation conducted within the Department of Biomedical and Clinical Sciences
"Luigi Sacco" it is clear that the technology Dust-Free FC UNIT 3″ has shown ability to lower the viral load of SARS-CoV-2 inoculated in liquid phase both on a surface and on a tissue.
Verified on cloth consisting of 45% polyether and 55% cellulose, inoculated with
SARS-CoV-2, exposed to treated air for 20 minutes in a volume of 2, 13 m3, showed instead a 2.5 log (99.7%) greater reduction than the natural decay of the virus.
VIEW TEST

Test ospedale dei colli
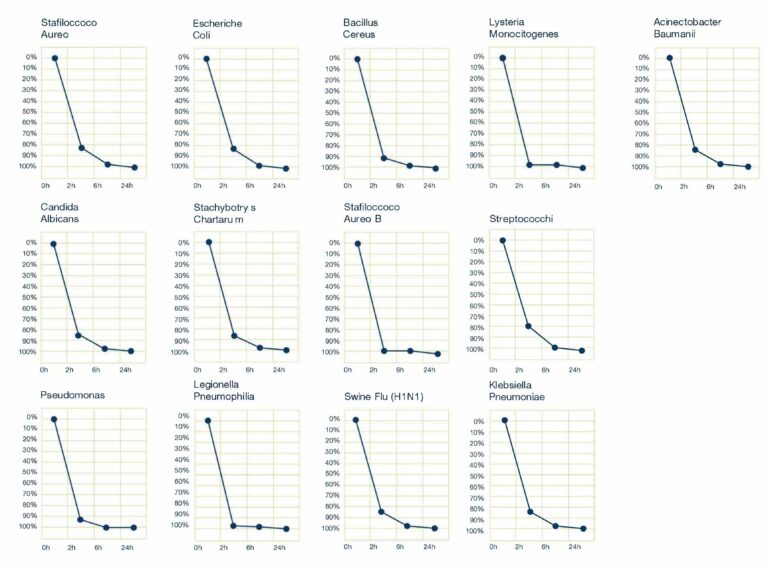
Test effettuato per verificare l’abbattimento dei seguenti virus e batteri:
Acinetobacter Baumannii MDR, Pseudomonas Aeruginosa, Klebsiella pneumoniae KPC, Staphylococcus aureus meticillino resistente.
VIEW TEST

Test formaldeide
L’indagine analitica sperimentale svolta non evidenzia formazione di sottoprodotto Formaldeide durante il normale funzionamento dell’apparecchiatura testata.
Le prove sono state effettuate in modalità controllata, tenendo conto dei parametri funzionali dell’apparecchiatura RefineAir PHOTOIONIX® ed in condizioni peggiorative in termini di accumulo e concentrazione delle eventuali sostanze sottoprodotti del trattamento (camera chiusa).
È possibile, pertanto, concludere che il dispositivo testato non determina condizioni di contaminazione dell’aria con sottoprodotto Formaldeide.
VIEW TEST

Test emissioni ozono
I test effettuati presso il laboratorio ProLab indicano emissioni di ozono quasi inesistenti
Visualizza test Photo-Family
Visualizza test Little Camp

EVALUATION OF THE BIOCIDAL ACTIVITY OF THE DUST FREE PROCESS with METHOD BASED ON EN 17272
Under the test conditions described, the DUST FREE process induces a reduction in the number of viable Staphylococcus aureus CIP 4.83 cells shed on stainless steel substrate by 1.3 to 2.2 log10 (i.e. 95% to 99% of the initial load) after a contact time of 6 hours at 20°C.
VIEW TEST - ACTIVE -
VIEW TEST - AIR KNIGHT -

DUST FREE MICROBIOLOGY TEST
Microbiological tests carried out by the American laboratory AirMid
VIEW

TEST ANTIMICROBICITA' COATING SU FILTRI
In questa prova, come campioni, sono stati utilizzati due diversi tipi di filtri, in due diversi sistemi di condizionamento per l’aria, dove si presume ci sia la presenza di un’elevata carica microbica.
I filtri testati sono:
• Filtro assoluto HEPA
• Filtro antipolvere Split
Per ciascun tipo di filtro di cui sopra, si e proweduto a testare un campione tal quale, senza trattamento, ed uno trattato con il coating, oggetto della valutazione.
I sistemi di condizionamento sono stati, quindi, contemporaneamente in funzione per un periodo di 4 mesi da giugno a settembre.
VIEW TEST

TEST EFFICACIA DISINFETTANTE REFINEAIR VS BOTRITE
I test effettuati hanno dimostrato la positiva efficacia del disinfettante
della RefineAir nel contrastare Botrytis cinerea, evidenziando una netta riduzione della crescita fungina e una marcata inattivazione del patogeno sulle superfici trattate.
Alla luce di questi risultati, l’utilizzo di tali disinfettanti risulta particolarmente promettente in ambito florovivaistico.
Un’applicazione ottimale potrebbe avvenire tramite sistemi di nebulizzazione automatizzati all’interno delle serre, nelle celle di conservazione e/o nel trasporto, in modo da garantire una distribuzione omogenea del prodotto, ridurre il rischio di contaminazione e contribuire in maniera efficace alla prevenzione e al controllo delle infezioni fungine.
VIEW TEST

University of Salerno microbiology test
Test microbiologico di microrganismi prescelti in capsule Petri, condotto in locale chiuso con cubatura di circa 35m3. La carica microbica depositata sulle piastre contenenti terreno agarizzato durante il trattamento con PCO-001 è nettamente inferiore a quella che si deposita quando nell’ambiente non è presente il dispositivo.
La differenza di contaminazione tra il campione ed il controllo è risultata tanto più marcata quanto maggiore era il tempo di esposizione delle piastre.
É possibile stimare che dopo 24 ore di trattamento la carica microbica (trattasi in prevalenza di muffe normalmente presenti nell’ambiente) riscontrata sul terreno agarizzato in presenza di PCO-001 è circa 100 volte inferiore (2 log) a quella del controllo cioè a quella presente su di una piastra lasciata nello stesso ambiente ma non in presenza dei nostri dispositivi di sanificazione dell’aria.
VIEW

TEST OF THE POSSIBLE RELEASE OF PHOTOCATALYST POWDER FROM THE SUPPORT DURING THE EXPERIMENTS

LEGIONELLA PNEUMOPHILA MICROBIOLOGY TEST
Microbiological testing on Legionella pneumophila abatement performed by MicroLife laboratory
VIEW

S.Epidermidis
The purpose of this report is to evaluate the impact of the Air Knight IPG by Dust Free in-duct air cleaning device on S.epidermidis levels in chamber air.
VIEW TEST

abbattimento formaldeide

Certificazione Ambientale - Settore Avicolo
Scopo del presente lavoro è stato quello di misurare l’efficacia del sistema di trattamento dell’aria in un ambiente naturalmente contaminato.
Per il campionamento dell’aria è stato utilizzato l’approccio attivo, tramite un flusso prodotto da un sistema di aspirazione, dotato di definizione della portata e dei volumi di aria.
VIEW

NEOTES LAB CERTIFICATE OF COMPLIANCE
The effectiveness of the sanitization actions have been tested in recent years by the accredited chemical and microbiological analysis laboratory Neotes, recording significant and encouraging results in terms of the reduction of microbial loads and of the main indoor pollutants.
VIEW

TEST UNIVERSITY OF GASTRONOMIC SCIENCES OF POLLENZO
The university of gastronomic sciences uses quality raw materials from teaching garden and from selected companies to offer students tasty and sustainable dishes. To reduce food waste at the source students and staff choose their meals every day through an online reservation system. In this test you can see the improved shelf-life of food
VIEW

EFFECTIVENESS OF THE DEVICE ON GASEOUS SUBSTANCES AND AIRBORNE MICROBIOLOGICAL AGENTS
Evaluation of the effectiveness of the device on gaseous substances (ammonia and methane) and aerodispersed microbiological agents present in the milking room at the zootechnical company Verdesca delle Fiocche dei F.lli Del Grosso in Fiocche di Eboli (SA).
VIEW

TEST MOCA
CNR TEST WITH CHIU-P SYSTEM
The tests were carried out by evaluating the performance of the system against fungal alteration due to Aspergillus niger, which is the main cause of deterioration of this product during commercial and domestic storage that which occurs in room temperature conditions.
VIEW
COMPENDIUM FOR DENTAL OFFICES
The Plus version of RefineAir sanitizers combines photocatalytic technology with bipolar ionization, increasing the quantity of positive and negative ions, making room air sanitization even more effective. In addition, the Little Camp and Photoionix models of RefineAir, from the air intake are equipped with a special patented titanium dioxide filter (spin-off affiliated with the Interuniversity Consortium for Materials Science and Technology INSTM), whose patent was obtained together with the Milan Polytechnic and Solari EMC in the "high technology bacterial purifier" area.
VIEW

British Insititute Microbiology test
The activity to study the effectiveness of the Refineair Device consists of monitoring the microbiological origin of air pollution in indoor environments before and after the installation of the RefineAir system.
The measurements carried out showed a significant reduction in pollution of microbiological origin with a percentage reduction of 69.5% for the total bacterial count at 22°, 59.1% for the total bacterial count at 37° and 70.7% for the yeast and mould count.
VIEW

Public transport tests
The test shows a reduction in exposure to pollution of microbiological origin with an average abatement percentage of 81.5% (microorganism count at 30°C), of 100% for yeast and mould counts.
VIEW

Campolongo Hospital microbiology test
The test involves microbiological monitoring on tools. The Data captured are pre- and post use of our active air and surface sanitization systems.
VIEW

TEST PERFORMED AT A DENTAL OFFICE
The Data captured are pre- and post use of our active air and surface sanitization systems.